|
It seems apparent that the seed moisture for storage is most critical to maintaining high germination percentage and initial root and shoot growth. Temperature was a factor but moisture content in storage was critical. Seed producers are aware of this concept and use many methods to monitor drying of seed after harvest, applying seed treatments and storing the seed.
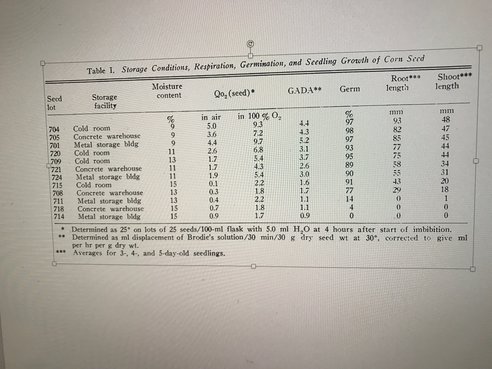
Higher moisture content in the seed activates some metabolism but not insufficiently to germinate effectively, causing an aging process. The pericarp does help protect air moisture diffusion into the kernel but genotypes vary in this pericarp feature, potentially complicating the drying and storage of the seed. Each individual kernel being slightly different physiologically and structurally contributes to the difficulty in producing and maintaining high percentage germination quality until planting time. Plant Physiol. 42, 1071-1076) compared seed respiration during imbibition, storage facility conditions, germination % and seedling growth of corn seed. Following table from that publication summarizes the interaction. Corn seed’s ability to rapidly grow new cells for shoots and roots after imbibing water deteriorates eventually. Unfortunately, this does not occur at the same rate for each seed within a seed lot or for all seed lots. Genetics, field history during seed production, and handling and environment of seed after harvest all affect the rate of deterioration for each individual seed. Associated with the deterioration is a reduced respiratory rate, consistent with the view that the mitochondria, where cellular respiration occurs, are not functioning properly. This process involves the membranes of mitochondria and probably are the main site of the problem. Each living cell includes multiples of mitochondria, each able to replicate and each able to repair membrane damage. Some of that damage occurred during the swelling from water imbibition. It is not difficult to imagine that as cells deteriorate, fewer mitochondria are able to duplicate. This reduces the total conversion of glucose to the chemical energy form (ATP), needed for the cellular duplication necessary for growth shoots and roots. Differences among the seed in this level of damage results in some being slower than others.
Mitochondria are an interesting part of the story of seed deterioration and cell function in all plants and animals. They are the size of bacteria. They can be seen with a light microscope but the anatomical details only seen with electron microscopes. They are the site of conversion of stored energy glucose to useable ATP energy needed for all other cell functions. More information on mitochondria was reviewed in the corn journal blog on 2/9/2016 http://www.cornjournal.com/corn-journal/mitochondria. One researcher’s personal experience with mitochondria research as it applies to humans is described at https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4137050. It is interesting and appropriate that this person grew up on a farm, where curiosity in biology can draw one deeper into trying to understand its basic dynamics. The fact that all individual seeds in a seed bag or a seed lot are not identical in terms of seed quality presents a problem for seed testing and seed companies. This problem gets greater as the more of the seed deteriorates as it gets difficult to judge those that do manage to push out the root and shoot from the seed. The ‘official’ practice is to designate those with torn or misshapen coleoptile or first leaf are called abnormals, and are usually equated as non-germinating in some labs. This is done under the assumption that these plants will emerge late or not at all when in the field.
Warm test methods vary, even with attempts to have similarity in materials, temperatures, watering and counting. Referee samples sent to 30+ public and private labs all testing seed from the same commercial bag of seed show a range of percent germinations of perhaps 5% when the average is high and 10% when the average among the labs is passing but below 95%. To accommodate this lab to lab variance, the legal range in actual germination printed on the tag on a bag of seed is 6% if the tag states 95% germination. Logical, systematic sampling of a seed lot and earnest efforts to test via the warm test still struggles to measure every seed’s germination quality. Cold test germinations are generally the most consistent predictor of emergence in the field. This seems to apply at least in the temperate zone because corn is generally planted soon after soil temperatures are higher than 50°F. The principle of cold test is that when seed imbibes but is unable to repair damaged membranes, the seed will either die, or not germinate with the non-damaged seed. Methods for cold tests vary more among labs as each one attempts to mimic field conditions, many by adding soil to the test. This can cause some problem because the water holding capacity affects oxygen available for respiration in the seed. PSR Inc. uses an artificial soil mix with a surfactant to minimize this problem. The many slight differences in methods among labs adds to the range of results that shows when each lab tests referee samples. Ultimately a seed producer needs to choose the lab and procedure that most consistently predicts the genetic potential of their product that will not be affected by seed quality. And this is not easy. This spring, living seed of slightly varying germination quality will be placed in varying environments with everyone’s intent to get maximum performance by the end of summer. Surely, testing of inanimate, manufactured objects instead of biological living things like seed is more defined but trying to understand the interactive dynamics of corn in its environments is more fun! Many studies and observations have supported the concept that uneven field emergence causes some reduction in final yield. There can be many causes of uneven field emergence, including moisture distribution, temperature, tillage, debris, pathogens, insects and seed germination quality. The problem is worst when the late emerging plants are adjacent to earlier emerging plants because of competition for root space, mineral absorption and light. Late emerging plants also push out silks later perhaps partly because reduced root volume fails to withdraw enough water to keep the silk cells from expanding at the normal rate. Late emergence of silks can come after most pollen from nearby plants has expired. At minimum, this can result in ear tips not filling but also can result in barren plants.
It is not always clear why germinations are not uniform in some seedlots. It appears to involve some aspect of membrane repair from aging or other deterioration of cell components. This supported by the observation that more uneven germination occurs after the seed is exposed to 7 days of 50°F before warmed to 70°F compared to the same seed sample placed only at 70°F. Given that each individual seed can have a slightly different physiological status, the slow emergers perhaps take a longer time to gain enough physiological strength to push the shoot above the soil surface. The complexity of seed quality status, and difficulty of precisely measuring it, plus the multitude of field interactions does not make it easy to predict the problems. Two reports that verify the reduction of yields from uneven emergence are: http://corn.agronomy.wisc.edu/Pubs/PC_JournalArticles/031-03-0811.pdf https://www.cornstates.com/News/NewsDocuments/ImpactofUnevenEmergenceinCorn-Jan23-TDA.pdf Also, this Corn Journal blog of March 10, 2016 (http://www.cornjournal.com/corn-journal/uneven-corn-seedling-emergence) relates my personal experience with monitoring late emerging plants that not only impressed me with the importance of annotating late emerging plants in germination tests but also set me off on exploring a different understanding for occurrence of stalk rot (it was not the late emerging plants). It is sad that we, and corn seed, age. The mechanisms between us and them may be similar in that mitochondria are probably involved in all deteriorating living cells. These organelles which can number a few hundred in a cell, are the main sites for transformation of stored carbohydrates into useable energy for other cell functions. Mitochondria have their own DNA and are composed largely of membranes. Dehydrated seed results in mitochondria functioning at a very low level resulting in being unable to repair deteriorating membrane structures. While at very low kernel moisture levels (6-14%?) and cool temperatures (less than 50°F (?) further damage is limited. Precise moisture percentage and temperature for best storage of maize seed probably varies for genotype and seed condition but the general concept remains.
Seed imbibition of water is a physical phenomenon with little inhibition from the pericarp or seed coat. Seed treatments added to seed can slow down the imbibition, apparently giving the renewed mitochondrial function more time to repair damaged membranes. On the other hand, only increasing kernel moisture slightly can cause more membrane damage to occur, but not repaired. Increasing moisture more while at low temperatures (50°F) has the same effect. Corn seed planted in cold soils will imbibe water but the low temperature inhibits normal cell function, including repairing mitochondria. Those individual seeds with the most mitochondrial damage are likely the ones that struggle to germinate when the soil temperatures do heat up. Seed producers are aware of the significance of inadvertently adding a small amount of moisture, such as from a seed treater before bagging by designing their process to limit the water and allowing for drying after application. I recall a case in the Thailand in which a new fungicide seed treatment was applied to control downy mildews but the humid environment did not allow the seed to dry after application. Seed germination quality quickly deteriorated as a result. Accelerated aging test of corn seed is based upon placing seed in an environment of 100% humidity and 113°F for 3 days, then planting in germination test to record the reduced germination. It is intended to predict the viability of the seed after storage. It is notable that even under this condition, all seed within the sample are not equally affected. Some germinated normally, some eventually and some not at all. This is typical of normal, well treated aging seed lots. Each individual seed is in a slightly different condition. We expect maximum performance when emergence is uniform. Seed quality is a factor influencing this trait. We all want things to be simple but it is amazing how rarely this happens. Chromosomal DNA from both parents of a hybrid influence the early growth of a germinating corn seed. However, the female parent has more affect then the male. It begins at pollination, in which the ovule is encased in the ovary wall destined to be the pericarp of the grain, making it a fruit with a single seed. This is totally composed of the cells of the female plant. Ovule cells of the female divide 8 haploid cells, one of which is the egg cell and 2 that become ‘polar cells’. Pollen contributes two haploid sperm nuclei, one fusing with the egg cell nucleus leading to the diploid embryo development and one fusing with the two polar nuclei and eventual formation of the triploid endosperm. The male contributed chromosomal DNA but the female parent also contributed important cell organelles such as mitochondria and plastids, including their DNA. The female parent contributes structure of the pericarp, two-thirds of the chromosomal DNA of the endosperm and the two important components of the cells.
Stress from drought, high temperatures or reduced photosynthesis during early days of grain fill are often associated with reduced germination percentage of maize seed. There is evidence that these stresses affect the balance between cytokinins and abscisic acid, the latter remaining at a high concentration and consequently reduced cell division in the developing endosperm. This results in fewer cells in the endosperm for storage of starch, affecting seed size but the effect on germination is not clear. Does shrinkage of endosperm put stresses on the pericarp, resulting in damage to the outermost layer of the corn kernel and consequential rapid imbibition? Mitochondria in the endosperm and embryo cells can withstand considerable drying stress, as witnessed by the effect of rapid drying after harvest. These organelles maintain life in the dehydrated state until activated with imbibition of water during germination. This is essential for them to provide chemical energy stored in the endosperm into growth of new cells in the embryo. Mitochondrial function is dependent upon membrane activity. Damaged membranes can repair under warmer conditions but not when soil temperatures are cold. Rapid imbibing of water can cause damage to membranes. Physical damage to kernel during harvest or after drying can damage the pericarp and other seed cells, resulting in fast imbibition as well. Hybrid chromosomal DNA affects many aspects of germination but female components are predominant, before and after harvest, on the vulnerability of the seed to germination. Potentials for producing less-than perfect seed are many. The art and skill of seed producers and the many others involved in maximizing the conversion of an ancient plant like Teosinte into an efficient converter of sunlight into useable form of chemical energy is amazing. Factors involved in producing the highest percentage and most uniform germination are understood by all experienced corn seed producers. Genetics, especially of the female parent, harvesting at appropriate grain moisture, careful physical handling from harvest, drying and shelling and bagging and proper storage are all important. Knowing the principles is significant and surely for manufacturing of most non-living products in controlled factories this would be adequate. However, genetics among hybrid parents vary and are selected based on many factors include hybrid yield. Although the date of optimum harvest is predictable, based upon pollination date, a weather front could cause considerable delay in harvest. It is established that moving volumes of dry air at temperatures around 90°-100F to quickly dry seed to about 12% gives the best germination. However, if outside humidity is close to 100% because of a series of rainy days, this is not always possible. Most producers can control the handling of the seed and storage of seed at proper temperatures and humidity before delivering to the distributers and farms.
Application of the principles of good seed germination quality requires experienced and careful management, not unlike most other aspects of agriculture. Success often depends on making the best decisions on when to harvest and adjusting drying bins according to the outside weather. PSR Inc. has been germinating commercial seed produced around the USA for 30 years. Low germination seed quality is usually associated with region of the country within a year, the region varying annually. This does not reflect any specific company but more that the environment of that season. Stress after pollination and harvest weather are major factors that can overcome the best of seed producer’s skills in some years. There are seasons in which the best knowledge, experience and decision making cannot avoid the detrimental effects of the environment on some seed lots. Fortunately, many of these stresses are temporary and very rarely affect all seed lots drastically at one location and rarely at any location on successive years. There is an art to producing high quality seed that adjusts to the environment of a season. The frequent success rate is indicative of application of the seed producer’s art. Perhaps, for the first 8 centuries of corn culture, the percent of seedlings emerging from seed planted was not as significant as the amount and type of grain produced. Planting in hills or even by planters at low densities, favored genetics that produced larger ears if adjacent seed did not germinate. Modern terminology attempts to identify hybrids that adjust of lower density by producing larger ears defining them as ‘flex’ ear types. It is doubtful that there are only two types, flex and non-flex, but hybrids do have different tendencies when plants are less dense. There are hybrids that will tend to go barren if planted too dense and there are others that will only show competitive yields if planted at high densities. There are some that do not cut back on kernel numbers if crowded but will develop high percentage of stalk rot if planted too thick. Recent years of corn culture in the USA has led to hybrids that tolerate planting a 33% higher density than 40 years ago- and give more stable high yields than hybrids of that era.
These modern hybrids may have genetics for more photosynthesis per acre, at least due to increased leaf area, but also consistent silking when under the stress from competing plants. This may also favor selection of hybrids with less ‘flex’ and consequently a need for higher plant density. Although best knowledge of the ideal plant density in any season only becomes apparent after harvest, there is more pressure now than 40 years ago to have a uniform emergence percentage of 90+% every season. Every seed within a single cross hybrid may be nearly genetically identical, but not with identical germination quality and not planted in identical microenvironments. Highest quality seed will tend to emerge uniformly and seed producers make large efforts to produce such seed. However, even with best genetics, timely seed harvest and care in handling the seed it is rare that field emergence is perfect. It is a challenge to produce all seed lots with great quality and for testing systems to correctly predict the field emergence the following spring. The large Cottonwood (Populus deltoides) tree close to our home covers our neighbors and our lawns, and fields with seed in late spring. A white oak (Quercus alba) tree produces huge volumes of acorns to be carried away by chipmunks and squirrels. Walnuts are buried by squirrels in late fall. A white paper birch (Betula papyrifera) covers a nearby deck with seed during the summer. Most flowering plant species have survived by spending reproductive energy in producing tremendous numbers of seeds, allowing a few to be positioned where they can germinate and established the next generation of the species. This strategy is not foreign to any one attempting to control weeds in a corn field- or a garden. Plant species have evolved many variants of this basic method leading to the wonderful plant variability, but this is not always advantageous to those of us interested in growing food.
Humans attempt to manipulate seed selection to not only propagate the species but also utilize the seed for their use. The original Teosinte species, from which corn was derived, had a hard case and very little endosperm for starch storage. People selected for softer outer case and much more starch storage in the endosperm. A more floury and softer endosperm became associated with a reduction in germination percentages, perhaps because of vulnerability to mechanical damage or perhaps faster imbibition damaging membranes. As corn culture moved to different geographies and selected for different food uses, seed functionality was affected as well. Corn seed breeders have the basic objectives of selecting genetics for use of the kernel, field performance of the hybrid and high germination percentage. It is good that corn has a very large number of genes to manipulate but also there are many physiological and structural aspects to corn seed germination. Whereas wild plant species mostly have developed reproduction strategies emphasizing large numbers of seeds to be widely distributed with a small probability of successfully producing the next generation, we have the dual objective of producing seed for our uses as well as a high probability of reproduction. Selection for both of these characters requires a lot of human effort |
About Corn JournalThe purpose of this blog is to share perspectives of the biology of corn, its seed and diseases in a mix of technical and not so technical terms with all who are interested in this major crop. With more technical references to any of the topics easily available on the web with a search of key words, the blog will rarely cite references but will attempt to be accurate. Comments are welcome but will be screened before publishing. Comments and questions directed to the author by emails are encouraged.
Archives
December 2021
Categories
|
 RSS Feed
RSS Feed