|
Every year there seems to be a newly found corn disease, or at least one that has been found for the first time in some corn-growing region. Given the wide range of environments, corn genetics, pathogen genetics and changing seasons this should not surprise us. Yet, when it occurs, everyone concerned with corn needs to try to understand the significance of the ‘new’ disease.
The 4th edition of Compendium of corn Diseases published by The American Phytopathological Society in 2016, covers at least 100 corn diseases plus lists multiple pathogens for some of the diseases. Many of these were prominent for a few years but changes in host genetics made them less significant only to later become notable again as susceptible genetics were used, or environments changed or the pathogen genetics changed. Goss’ wilt surprised us in the early 70’s, then became much less significant for most dent corn as resistant hybrids became common. But, during the last few years this disease has become damaging again. Did the pathogen change or is this totally because of use of susceptible hybrids? Gray leaf spot was known as insignificant until about 40 yrs. ago, and then only in the humid, foggy eastern USA valleys. A few years later, it became notable in the river valleys of the Midwest and then we discovered it likes the irrigated fields of the western Midwest. On the other hand, sorghum downy mildew had a spurt of attention in the 70’s in which It overwintered on Johnson grass, increased on sorghum and then went to corn in areas of Missouri and Kansas and south. Better herbicide control of the Johnson grass broke the cycle, greatly limiting the destruction from this disease. It is probable that new corn disease dynamics are occurring somewhere each year but undiscovered until the disease increases to an extent that it is noticeable and spread over some area. Recently there were announcements concerning bacterial leaf streak, caused by Xanthomonas vasicola pv. vasculorum, occurring in Midwestern fields. Early symptoms could be confused with gray leaf spot and thus far it has not been identified with damage to performance to the crop. We do not know if it will be damaging, how it spreads (it is not believed to be seed borne), does it have other corn belt hosts (it is also found on sugar cane in Africa), is it attacking only some genetics or is it favored by certain environments. It was first reported in 2015 in Nebraska. Public and private researchers are trying to learn more about this disease and it is probable that as the dynamics are understood, this disease will join the many others that occur but are insignificant to corn production. The first role for a corn pathologist involved with a new corn disease is to identify the pathogen but the most important, and most difficult, is to predict the significance of the disease. There are genetics, of course, behind the chemical responses to attacks by pathogens. The genetics must relate to detection of an attack, perhaps detection of the specific pathogen, production of a general or specific anti-pathogen material and speed of the response.
Although all resistance to corn diseases involves multiple genes affecting the biology for the processes mentioned above, in many diseases there is a single corn gene that has a drastic, critical affect on the pathogen. This is called qualitative resistance. In the case of rust diseases, the pathogen is killed almost as quickly as it invades a cell. With Exserohilum turcicum the plant with an Ht gene stops the fungus as it enters the vascular system, inhibiting the fungus from producing usual northern leaf blight lesions and spores to further spread the disease. Qualitative resistance is convenient for the corn breeder to select during the breeding process but, unfortunately, nearly always the population of the pathogen includes individuals with a single gene that produces a product to overcome the resistance product. Eventually, natural selection results in increasing presence of those pathogens with this gene and the the corn single gene is less useful. The more stable resistance involves strengthening the detection, and the speed and concentration of the anti-pathogen materials. This is called quantitative resistance. It is controlled by several genes and therefore more difficult to identify by breeders. It is usually reflected by fewer and smaller lesions but assurance of exposure to the pathogen comparable to potential for the commercial life of the hybrid is not simple. Most diseases are heavily influenced by environments which vary by location and season. Most susceptible genetics are eliminated in breeding nurseries but we do get surprised occasionally either with increased intensity of a disease or new pathogen changes. Disease resistance in plants is not totally unlike that in humans. A pathogen is detected, the host responds by producing hormones that ultimately result in production of pathogen-inhibiting chemicals that eventually stop the pathogen.
Corn leaf epidermal cells are tightly connected except for the stomata. The vast majority of microbes surrounding corn plants cannot penetrate the plants. The few that do have the capability, perhaps by enzymatically drilling through the epidermal cells to enter the leaf tissue, set off the alarm. In some cases, the plant’s first response is increasing the production of salicylic acid in the area of the invasion. This turns on the genes for production of the protein, often an enzyme, with the capacity of stopping the pathogen from spreading. This final product may be effective against several potential pathogens or specific to one species. Our company, Professional Seed Research Inc., offers a service of evaluating disease resistance in experimental corn hybrids for seed companies. We culture the pathogens and then inoculate the plants to the fungal pathogens by applying a concentration of spores to the whorls of plants when at about V7. It is common that this moist environment assures spore germination and infection in the leaf tissue that was in the whorl at the time. We inoculate only one pathogen per plant to avoid the resistance mechanism triggered by another pathogen and therefore not representing the genetics of the hybrid. A rain storm arrived the evening after we inoculated the nursery in 2016. Symptoms for the southern corn leaf blight and northern corn leaf blight were very sparse on the respectively inoculated plants. Instead there were common rust pustules in the areas of the leaves that should have shown the other two diseases. The hypothesis is that the rust spores were distributed by the storm into the leaf whorls, and the rust fungus (Puccinia sorghi) infected the leaf tissue. This triggered a general resistance factor that inhibited the other two fungi (Bipolaris maydis and Exserohilum turcicum). I recall this happening several years ago as well. There is a published report of infection by either of the latter two fungi limiting the infection by the rust fungus. Hopefully, in our case, inoculating much later with the intended pathogens will find that the general resistance factor will no longer be present. Among the aspects of corn biology that I find fascinating is the application of knowledge of a single plant to growing the crop. Corn is a grain crop and we use it primarily to convert light energy into chemical energy in the form of carbohydrates. We know that photosynthesis in corn increases with light intensity striking the leaf. We also know that corn varieties differ in leaf structure, allowing differing amounts of light to penetrate the canopy. Measurements of leaf area index (LAI) can be made to compare effective photosynthetic leaf areas among hybrids, plant densities, fertilizer applications and and plant maturities. We also can measure the rate of photosynthesis per unit of leaf area among hybrids and fertilizers.
In addition to the the photosynthesis dynamics the effectiveness of pollination, kernel numbers establishment, kernel fill rates, leaf disease resistance, and root growth are among the inherited characters of a hybrid that contribute to its ultimate grain production. A recent study claimed that corn has 100,000 genes. We may know some of these individual genetics but ultimately it is the measurement from yield trials done by seed providers and then experienced by growers that determine the best converter of light to grain, at least for that individual field that year. Unfortunately, the best hybrid or plant density or fertilizer program for 2016 may not be the best for 2017. But we do know that corn is an outstanding converter of light energy into carbohydrates. 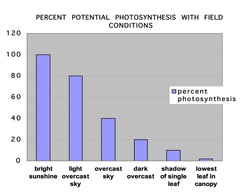 Our eyes don’t allow us to be aware of differences in light intensity but the photosynthesis rate in corn leaves is very sensitive to this factor. Having a C4 photosynthesis system, the rate of photosynthesis in an exposed chloroplast increases directly with light intensity up to full sunlight. C3 plants, such as soybeans, can only use 3/10ths of full sunlight hitting the leaves. Several years ago, I carried a light meter to measure foot candles in many corn environments to help me understand light intensity differences. Measuring in foot candles is not quite as good as PAR (Photosynthetically Active Radiation) measurements as done with more recent instruments but it did demonstrate to me the differences in light intensity within corn environments. Full, unobstructed sunlight has intensity of 10000 fc (foot- candles) when measured in Ohio, Illinois and in Colorado. However, the measurement in the shadow of a single corn leaf is only 1000 fc. That portion of the leaf in the shade of the single leaf is only producing 1/10th the carbohydrate as the same area of the leaf fully exposed to direct sunlight. The lowest leaf in a canopy receives only about 50fc, hardly enough to sustain its own tissue and is one of the reasons that these leaves disintegrate. As shown in the graph above, cloudy days also reduce the photosynthetic rate in corn plants. It is a precarious balance between sugar demands established in the ear after pollination and factors affecting photosynthesis in fields. Growers are in control of some factors such as hybrid selection, fertilizers and plant density but others such as rain affecting kernel numbers, or leaf diseases potentially removing effective leaf area and cloudy weather can drastically affect whether the corn plant stays alive until the completion of grainfill. As the corn embryo develops, and the cytokinins accumulate in the pollinated ovule after the first 10 days, there is a constant translocation of sugars to each kernel for each day. The total daily movement continues for about the next 40 days, almost regardless of daily variable rates of photosynthesis due to cloudy weather or leaf damage from disease. Sugars are drawn from all leaves and even those stored in the stalk pith tissue. The total draw to the ear is determined by genetics of the variety, environmental factors including minerals and the number of kernels. The number of kernels is also determined by genetics and environment factors such as minerals and especially water available during ovule formation and pollination.
The daily transfer of sugars during days 50 to 60 of grain fill is greatly reduced until the abscisic acid affect causes thick cell walls to form at the base of the kernel, cutting off the sugars transfer into the kernel and the movement of water from the kernel. This is known as the black layer. Sugars translocated to the ear are sugars not available to other living tissue in the plant. Roots are especially dependent on the same sugars to support metabolism functions, including warding off the potential microbe invaders. Starving roots, as they rot, eventually reduce water uptake and, if insufficient water to meet the transpiration rates from leaves, a permanent wilt will occur. With the wilt, movement of sugars to the kernels is stopped, abscisic acid takes over, causing the black layer to form a base of kernel. The consequence is light grain weight on the affected ear. Corn plant growth is greatly affected by a broad class of complex chemicals called hormones. Two of the kinds of hormones related to grain fill are the cytokinins and abscisic acid (ABA). These two hormones have opposing functions in plants, including the development of corn kernels. Cytokinins function is to increase cell division and delay senescense of tissue. They are produced in roots and transported via the xylem to meristems such as in each kernel. They also may be produced in seed embryos also but evidence for that is elusive. Regardless, cytokinins accumulate in developing seeds where they are responsible for stimulating cell division. Cytokinins are also linked with the transportation or at least the attraction of sugar to the developing kernels.
Abscisic acid, on the other hand, is associated with cutting off of translocation to tissue basically by causing a layer of thick walled cells impervious to movement of materials. Abscisic acid production increases when the plant is stressed. The black layer at the base of mature corn kernels and at the base of husk leaves in a mature corn ear are stimulated by abscisic acid. Freshly pollinated ovules have a balance of these two hormones. A non-stressed corn plant normally has a balance favoring the cytokinins stimulating more cell division and, consequently, flow of sugars to the individual kernels. However, if the plant is under heat or drought stress the balance tends to favor abscisic acid. The affect can be abortion of those kernels. Corn kernels within the first 10 days after pollination are most vulnerable, perhaps because the accumulation of cytokinin is too great to be overcome by a short-term increase in abscisic acid. Genetics and environments influence the production of these two critical hormones affecting grain yield in a corn field. Prominent ear rotting fungi such as Aspergillus flavus, Stenocarpella maydis (Diplodia), Fusarium verticilloides and Fusarium graminearum (Gibberella zeae) have multiple ways of attacking the developing ear. One of the means of attack is through the silk. Fusarium species have been shown to travel through the silk channel and through the micropyle into the pollinated ovule. Others have been shown to move on the surface of the silks to infect the developing kernel pericarp. If the pericarp and seed coat of the developing kernel is wounded by insects such as ear worm, the fungus gains entry into the kernel. Stenocarpella maydis can enter through silk, but often infects the base of the ear and surrounding husk tissue. From stem tissue at the base of ear it grows in the young developing ear. Aspergillus flavus, producer of aflatoxin, grows on the surface of silk and then on the surface of kernels, entering the kernels only with injury.
All of these fungi persist in soils but differ how they are favored with environments. Timing of rain seems to greatly affect the occurrence of the infection. There are varietal differences in resistance to each of them that can be measured in controlled conditions. Field evaluations based upon natural infection carries risks of faulty conclusions because stresses that cause delays in silking because of soil drainage differences, or maturity differences in relation to time of rain can be confusing. There are times when a variety may appear to be susceptible in one variety display plot but the same hybrid with different planting date would be judged as resistant in another plot. Caution is advised in drawing conclusions concerning resistance among corn hybrids. Ustilago maydis is the fungal pathogen of corn causing common smut disease. Its biology includes a fungal version of sex, a portion of which involves corn. The fungus produces thick walled spores called teliospores that overwinter in the soil for many years. Teliospores have diploid nuclei (2 sets of chromosomes). When moistened, the teliospore nuclei undergo meiosis, resulting in 4 individual cells, each with haploid (monoploid) with one set of chromosomes. These cells are called sporidia. Sporidia that land on corn tissue may germinate but cannot enter the corn plant until they are united with sporidia of a different mating type. After the two mating types fuse, the two nuclei stay separate but the fungus forms special structures to enter the corn plant cells. Often it is through wounds but also directly through corn cells as they are elongating, as the combination of host cells and fungus form galls. Within this mass the fungal cells, having two monoploid nuclei per cell, now fuse the nuclei, forming the diploid single nucleus stage of the fungus life cycle. These diploid cells form the thick cell walls as they become teliospores to be released as the corn plants are harvested.
Smut galls may form on leaves, tassels, aborted ear shoots at the leaf nodes and the main ear shoots. The ear shoots are the most damaging to the grain yield, of course. Corn silks that are not pollinated soon after emergence are vulnerable to infection by Ustilago maydis. The fungus grows down the silk channel infecting the cells surrounding the ovule. If pollen successfully reaches the ovule before the fungus, the smut fungus is inhibited and no gall will form. Subsequently, the timing of pollen release and silk emergence becomes a significant factor in formation of galls on corn ears. Rainy days may encourage silk emergence but delay pollen release or drought may delay silk emergence until there is limited pollen available. Complete absence of kernels but ears full of smut are a sign that the ear failed to receive pollen. These factors contribute also to the occurrence of smut at the tips of ears as these are from the last silks to emerge, perhaps when no pollen was available. I initially entitled this blog issue as ‘Smut, sex in corn’ but later decided it might be misleading to some search engine users. Readership could temporarily increase but some may not proceed beyond the first sentence. |
About Corn JournalThe purpose of this blog is to share perspectives of the biology of corn, its seed and diseases in a mix of technical and not so technical terms with all who are interested in this major crop. With more technical references to any of the topics easily available on the web with a search of key words, the blog will rarely cite references but will attempt to be accurate. Comments are welcome but will be screened before publishing. Comments and questions directed to the author by emails are encouraged.
Archives
December 2021
Categories
|
 RSS Feed
RSS Feed